Curiosity fuels us,
collaboration shapes us.
Wonder Research advances mental healthcare through FDA Clinical Studies in psychedelic science, novel protocol development, and academic partnerships.

The Wondermed Pilot - Completed
Four years of clinical data powering the AI system
Wondermed’s 46-month telemedicine platform generated real-world data now embedded in Wonder Sciences’ AI systems and protocols.
- 1,919 patients treated across real-world care settings
- 24 U.S. states with active provider deployment
- 40+ providers across a multi-state clinical network
- Mental healthcare needs a 360° AI operating system
- AI-native clinical operations generate exponential productivity
- One billion people are in need across a trillion-dollar market
- A chronic pain protocol would have the highest positive human impact
4 Advanced AI Utility Patents Filed, one unified, AI-native system with three integrated FDA products.


IRB Clinical Research Study with UCLA - Completed
“Sublingual Ketamine for Depression and Anxiety: A Retrospective Study of Real-World Clinical Outcomes”
UCLA IRB Study: 431 patients enrolled
- 89% of patients with moderate to severe depression experienced clinically meaningful symptom improvement
- 80% of patients with moderate to severe generalized anxiety disorder experienced clinically meaningful symptom improvement
- 84% of patients with moderate to severe social anxiety disorder experienced clinically meaningful symptom improvement
Reengineer protocol focused on the science of Flow State - FDA 505(b)(2)


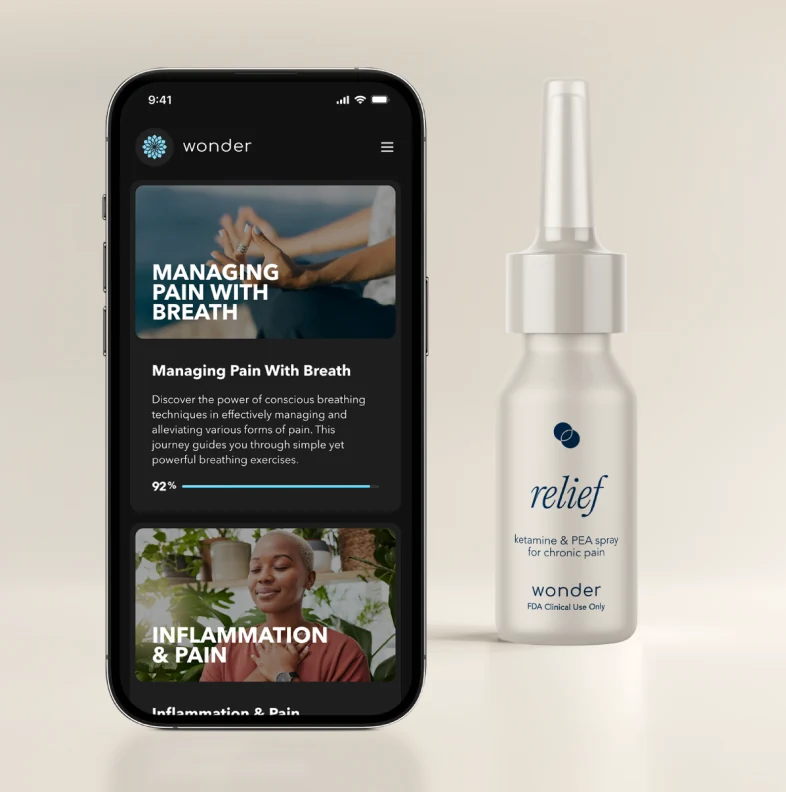
A safer alternative to opioids for chronic pain
- FDA 505(b)(2) Pathway
- Leading Academic Partner
- Study Launch: Q2 2027
- Target NDA: 2030
- 12-Week Virtual Protocol combining low-dose ketamine, supplements, CBT, mindfulness, and coaching
- AI-Assisted opioid tapering with human-in-the-loop clinical oversight via WondermedAI
- Continuous monitoring with real-time safety alerts via WondermedAI
- Proprietary AI abuse surveillance designed for patient safety and regulatory compliance
- Adaptive FDA Phase 2/3 design enabling accelerated clinical development
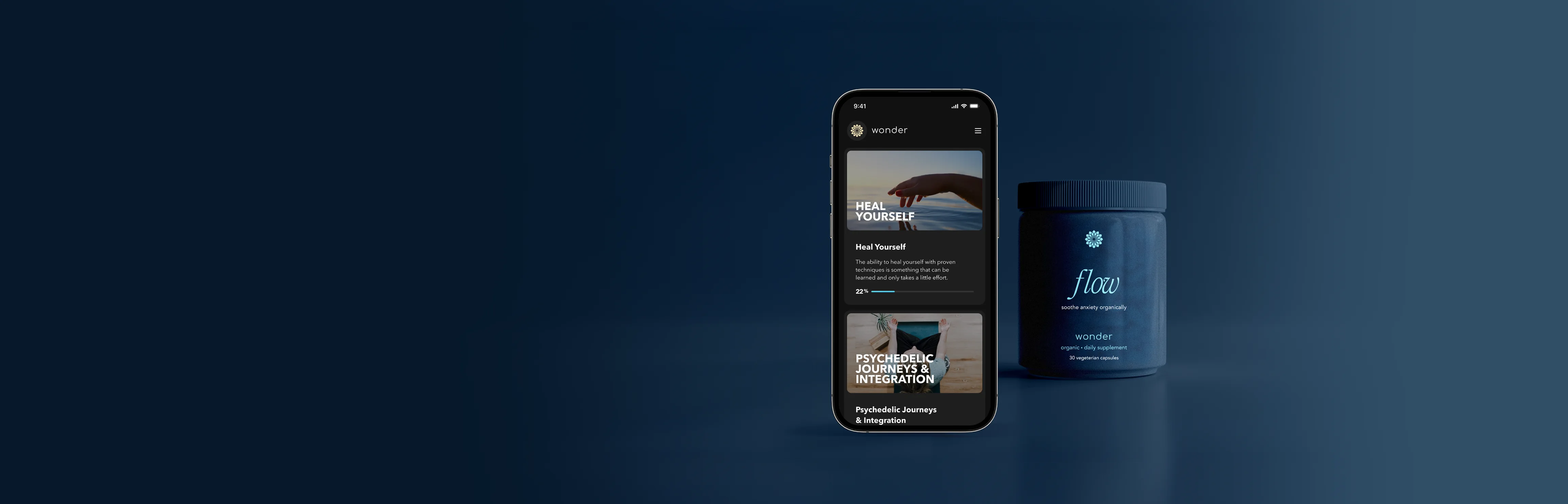
AI-guided protocol for anxiety and depression
- Flow State Protocol – Built on the neuroscience of flow to enhance neuroplasticity and therapeutic outcomes
- Wonder Ketamine Lozenges – Proprietary formulation paired with cannabinoids to optimize efficacy and tolerability
- WonderFlow Daily Supplement – Designed to support neuroplasticity, recovery, and treatment continuity
- AI Personalization – Adaptive dosing, timing, and intervention pathways powered by patient response
- Validated V1 Learnings – Refined from 1,500+ patients and IRB-backed clinical data
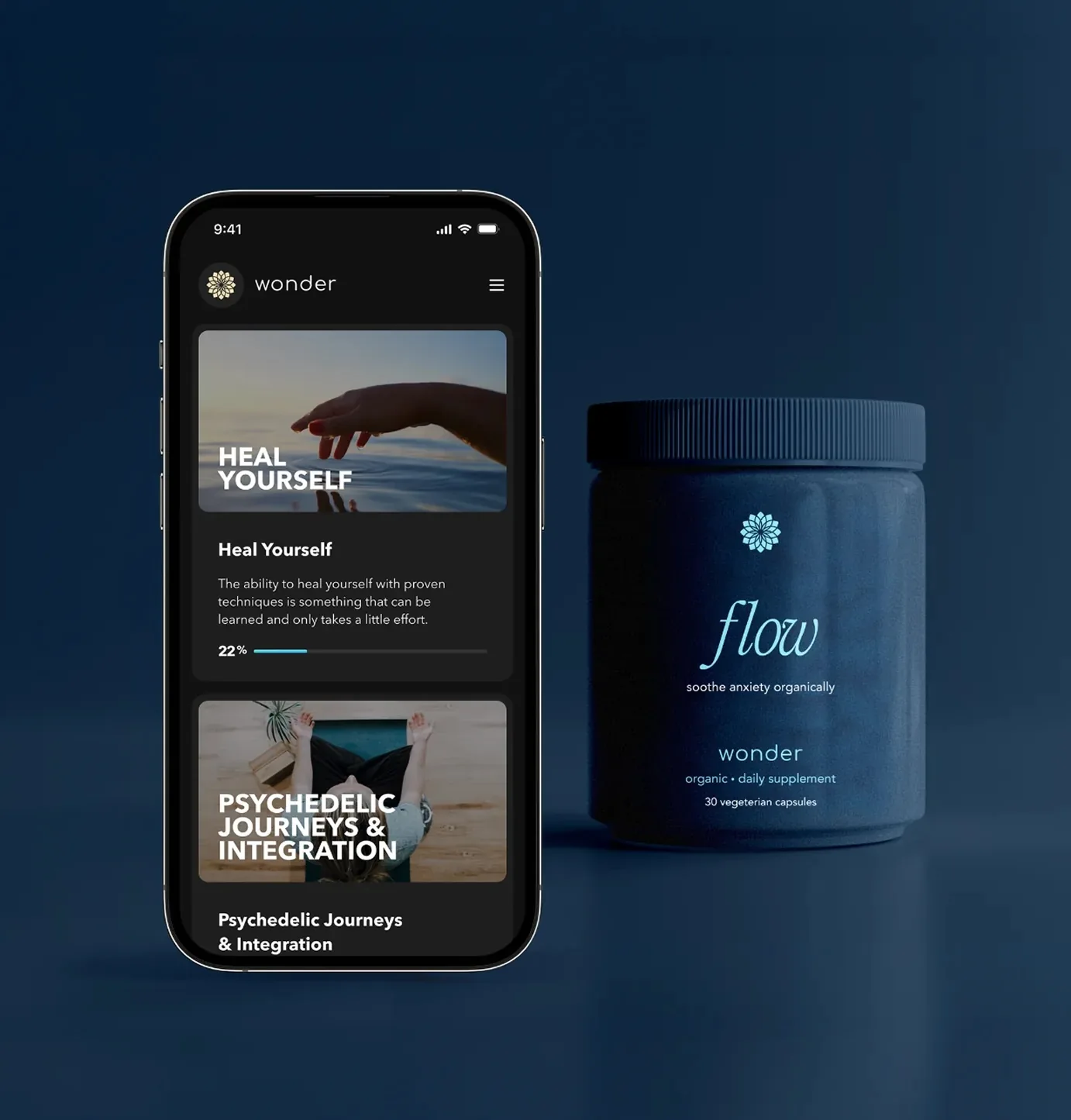
The AI-native clinical OS for precision mental healthcare
Target FDA Approval: Q4 2028
- WonderAI Pharmacology – Real-time prescribing intelligence and optimization across neuroplastic and psychiatric medications
- WonderMate Copilot – Unified clinical support for prescribers and therapists
- Wonder Cognitive (Cognitive Twin) – Continuous patient model across medication, therapy, and behavior
- Continuous Monitoring – Always-on tracking, alerts, and adaptive care
- AI-Native Infrastructure – One system connecting patients, providers, and protocols


collaboration shapes us.
